The National Advisory Committee on Immunization (NACI) had previously recommended third doses for immunocompromised people 12 years of age and older.

Children age five to 11 who are moderately to severely immunocompromised should get a third dose of COVID-19 vaccine, Dr. Theresa Tam, Canada’s chief public health officer, said on Friday.
The National Advisory Committee on Immunization (NACI) issued the updated recommendation on Tuesday, but Tam highlighted it in a COVID-19 briefing on Friday.
Last September, NACI recommended that people 12 years of age and over who are moderately to severely immunocompromised should receive three doses of the vaccine.
The third dose given to children and adolescents should be the Pfizer-BioNTech mRNA vaccine, the committee said, noting it should be given four to eight weeks after the second dose.
According to the latest NACI guidance, children and teens are considered immunocompromised if they have one of the following conditions:
- Active treatment for solid tumour or hematologic malignancies.
- Receipt of solid-organ transplant and taking immunosuppressive therapy.
- Receipt of chimeric antigen receptor (CAR)-T-cell therapy or stem cell transplant (within two years of transplantation or taking immunosuppression therapy).
- Moderate to severe primary immunodeficiency with associated humoral and/or cell-mediated immunodeficiency or immune dysregulation.
- Treatment of HIV with immunosuppressive therapies.
NACI emphasizes recommendation that children be vaccinated
NACI’s latest guidance also “strengthened” its previous recommendation that all children five to 11 years of age be vaccinated with two pediatric doses of the Pfizer-BioNTech vaccine. The pediatric dose is one-third (10 micrograms) the size of the 30-microgram dose given to adolescents 12 years of age and older and to adults.
WATCH | Will updated recommendation improve vaccine uptake for children?
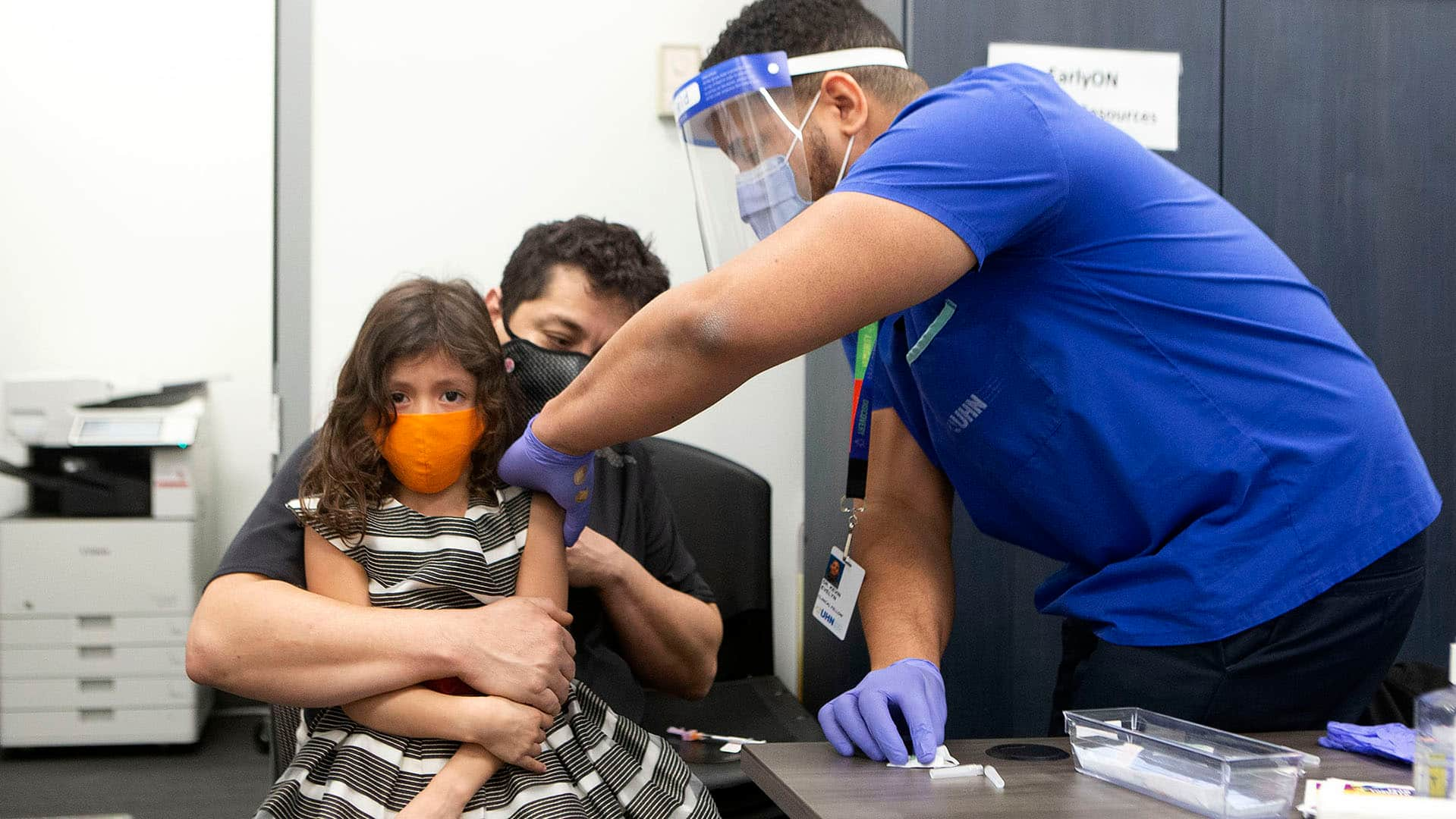
COVID-19: Will updated guidance improve vaccine uptake in kids?
Infectious diseases specialist Dr. Isaac Bogoch answers questions about whether the National Advisory Committee on Immunization’s guidance that children should get the COVID-19 vaccine will improve uptake and how concerned people should be about an emerging variant. 4:25
“Children five to 11 years of age continue to remain at low risk of severe COVID-19 outcomes, including from the Omicron variant; however, the number of children experiencing severe disease or requiring hospitalization is increasing due to [a] large number of children becoming infected with SARS-CoV-2 during this wave of the pandemic,” the updated NACI guidance said.
ABOUT THE AUTHOR
Nicole Ireland is a CBC News journalist with a special interest in health and social justice stories. Based in Toronto, she has lived and worked in Thunder Bay, Ont.; Iqaluit, Nunavut; and Beirut, Lebanon.
- @nicireland_news

