Citing numerous E. coli O157:H7 outbreaks linked to leafy greens, the FDA is telling the industry that it’s past time for a head-in-the-sand view of cattle operations uphill from lettuce fields.
In a new report, the Food and Drug Administration outlines what it wants to see the industry do and how it came to those conclusions. The report on the investigation of an October 2020 outbreak is the first of a one-two punch released today. The second hit involves updates to the FDA’s Leafy Greens Action Plan, first published in 2020.

Part of the action plan discusses what the FDA wants the produce and animal agriculture industries to do to reduce the ongoing dangers of contamination of foods generally consumed in their raw form. The same strains of E. Coli keep coming up in leafy greens outbreaks and those strains have been found in samples of animal droppings.
By far the most frequently referenced likely contamination sources in the FDA report and action plan are cattle operations located adjacent to and uphill from leafy greens fields. Wildlife habitats are also mentioned as problematic.
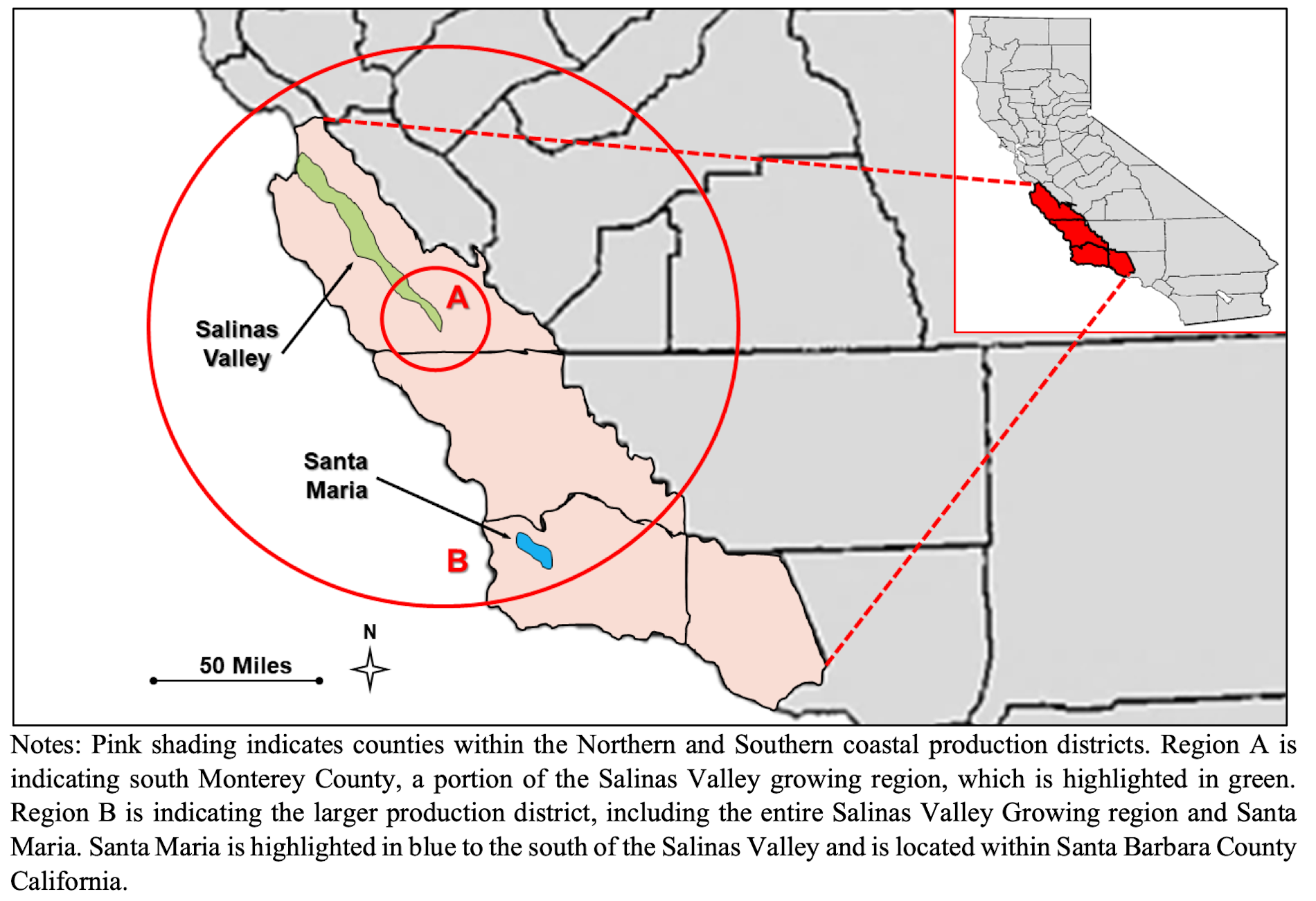
“This reoccurring pathogenic E. coli strain, therefore, appears to be a reasonably foreseeable hazard in the California Central Coast leafy greens growing region, and of specific concern in the South Monterey County area of the Salinas Valley growing area,” according to the FDA report.
“Farms subject to the FSMA Produce Safety Rule are required to take all measures reasonably necessary to identify, and not harvest, produce that is likely to be contaminated with a known or reasonably foreseeable hazard.”
“The updated plan includes a renewed emphasis on actions to prevent contamination stemming from activities on adjacent land. . .”
The FDA’s report acknowledged efforts by “locally-led” produce growers, livestock owners, and other agricultural industries including the Monterey County Farm Bureau, and CDFA. Their work should continue and broaden as both short-term and long-term solutions need to be pursued with urgency, the FDA says.
A cattle operation uphill from leafy greens fields has been examined as a possible source in more than one outbreak, according to the FDA investigation report, which focused on the October 2020 outbreak linked to romaine lettuce.
The report says outbreaks in 2017, 2018, 2019, and 2020 are not a new phenomenon.
“In the United States, E. coli O157 outbreaks were first linked to contaminated leafy greens in 1995. Later, studying a decade of investigations between 2009 and 2018, the FDA and Centers for Disease Control and Prevention (CDC) identified 40 foodborne outbreaks of STEC infections in the U.S. with a confirmed or suspected link to leafy greens,” according to the FDA’s updated Leafy Greens Action Plan.
“. . . recent investigation findings and previous foodborne illness outbreak investigation findings dating back to 2013 suggest that a likely contributing factor for pathogenic E. coli contamination of leafy greens has been the proximity of cattle. Cattle have been repeatedly demonstrated to be a persistent source of pathogenic E. coli including E. coli O157:H7. . .
“Continued outbreaks of this reoccurring strain of pathogenic E. coli associated with leafy greens consumption impose a significant public health burden, reduce consumers’ confidence in the food supply, and necessitate more collaboration by growers, livestock owners, and the broader agricultural community to improve response efforts and protect public health.”
Specific recommendations FDA wants industry in the California Central Coast Growing Region — encompassing the Salinas Valley and Santa Maria growing regions — to keep in mind include:
- Consider this particular strain of E. coli O157:H7 a reasonably foreseeable hazard. Under the Produce Safety Rule (21 CFR 112.112 and 112.113), farms must: take all measures reasonably necessary to identify, and not harvest, a covered product that is reasonably likely to be contaminated with a known or reasonably foreseeable hazard; and handle harvested covered produce during covered activities in a manner that protects against contamination with known or reasonably foreseeable hazards.
- When pathogens are identified through microbiological surveys, pre-harvest, or post-harvest testing of leafy greens, we recommend growers implement industry-led root cause analyses to determine how the contamination likely occurred and then implement appropriate prevention and verification measures.
- Actively engage in the locally-led, locally-convened effort called California Agricultural Neighbors (CAN) workgroup that is being led by the CDFA and Monterey County Farm Bureau to identify what actions can be taken to reduce the risk of STEC contamination of leafy greens in this specific growing region.
- Actively seek participation in the California Longitudinal Study (CALS) to better understand the ecology of human pathogens in the growing region. This multi-year effort is intended to provide information on how human pathogens survive in the broader agricultural environment and how they move throughout the environment, leading to potential produce contamination prior to or at harvest. The results from this collaboration are intended to lead to improved practices to prevent or mitigate food safety risks, and ultimately help enhance the safety of produce grown in the region.
- Actively encourage participation by adjacent and nearby livestock owners and other agricultural producers in the region to participate in CALS as well.
- Be aware of and consider risks that may be posed by adjacent land use, especially as it relates to the presence of livestock and the interface between farmland, rangeland, and other agricultural areas.
- Assess growing operations to ensure implementation of appropriate science- and risk-based preventive measures, including applicable provisions of the FSMA, Produce Safety Rule, and good agricultural practices.
- Increase digitization, interoperability, and standardization of traceability records along the entire leafy green farm-to-store continuum, which would expedite traceback and prevent further illnesses. This is important at not only the grower level, but critical for shippers, manufacturers, and retailers as well, to improve overall traceability throughout the supply chain.
A specific area of focus in the FDA’s documents is that of traceability.
“After the 2018 outbreak of E. coli associated with romaine lettuce, growers led a change in labeling to identify a growing region on packaged romaine lettuce. While better food traceability back to the farm is the ultimate goal, as an interim measure, provenance-labeling has been helpful in identifying during traceback investigations where potentially contaminated romaine was grown,” the agency report says.
“Thus, the industry should consider adoption of improved — with more detailed specificity beyond the current region designations — and expanded to leafy greens products beyond just romaine lettuce labeling to further protect consumers in the event of a product recall and minimize the amount of discarded product.”
The nature of leafy greens — being grown outside — makes it difficult to mitigate contamination dangers, according to the FDA, but those difficulties are no reason to give up the fight or accept the status quo. However, 2020 brought unique and unavoidable changes to the status quo, which the FDA says spurred part of the update to its Leafy Greens Action Plan.
“. . . 2020 presented unique challenges to implementing the action plan. The COVID-19 pandemic specifically made some actions difficult to accomplish, creating the need to continue some work with a renewed emphasis in the year ahead,” the FDA update says.
“. . . greater emphasis will be needed around such complex issues as adjacent land use, agricultural water, and understanding likely routes by which human pathogens may contaminate leafy greens.”
New recommendations in the 2021 Leafy Greens Action Plan include:
- Public meetings, workshops, reach out to chemical suppliers to remind them of EPA rules;
- Additional, focused sampling assignments for romaine lettuce grown in Arizona;
- Enhance communication and cooperation between government, industry, and academia, promptly releasing new investigation findings;
- Publish an additional review of FDA-investigated on-farm findings associated with outbreaks of Shiga toxin-producing Escherichia coli infections linked to romaine lettuce from 2009-2018;
- Finalize and post a document summarizing all actionable data for consideration in directing future policy, guidance, and research activities around the safe and effective use of biological soil amendments of animal origin.
Recommendations from the original version of the FDA’s Leafy Greens Action Plan published in 2020 that are listed in the updated plan include:
- Continue third-party audits;
- Create a voluntary public-private data trust for leafy greens;
- Continue working with stakeholders to share knowledge on new technologies;
- Continue ongoing dialogue on region-specific issues in real-time with a broad array of Yuma, AZ, growing region stakeholders;
- Advance the development of a final rule for implementing FSMA Section 204 related to the records required for tracking and tracing designated foods, which may serve as a foundation for traceability throughout the entire food system; and
- Continue work with retailers and government partners to improve the timely collection and transmission of purchase information during an open traceback investigation.
(To sign up for a free subscription to Food Safety News, click here.)

